|
9/25/2023 0 Comments Samtools get consensus sequences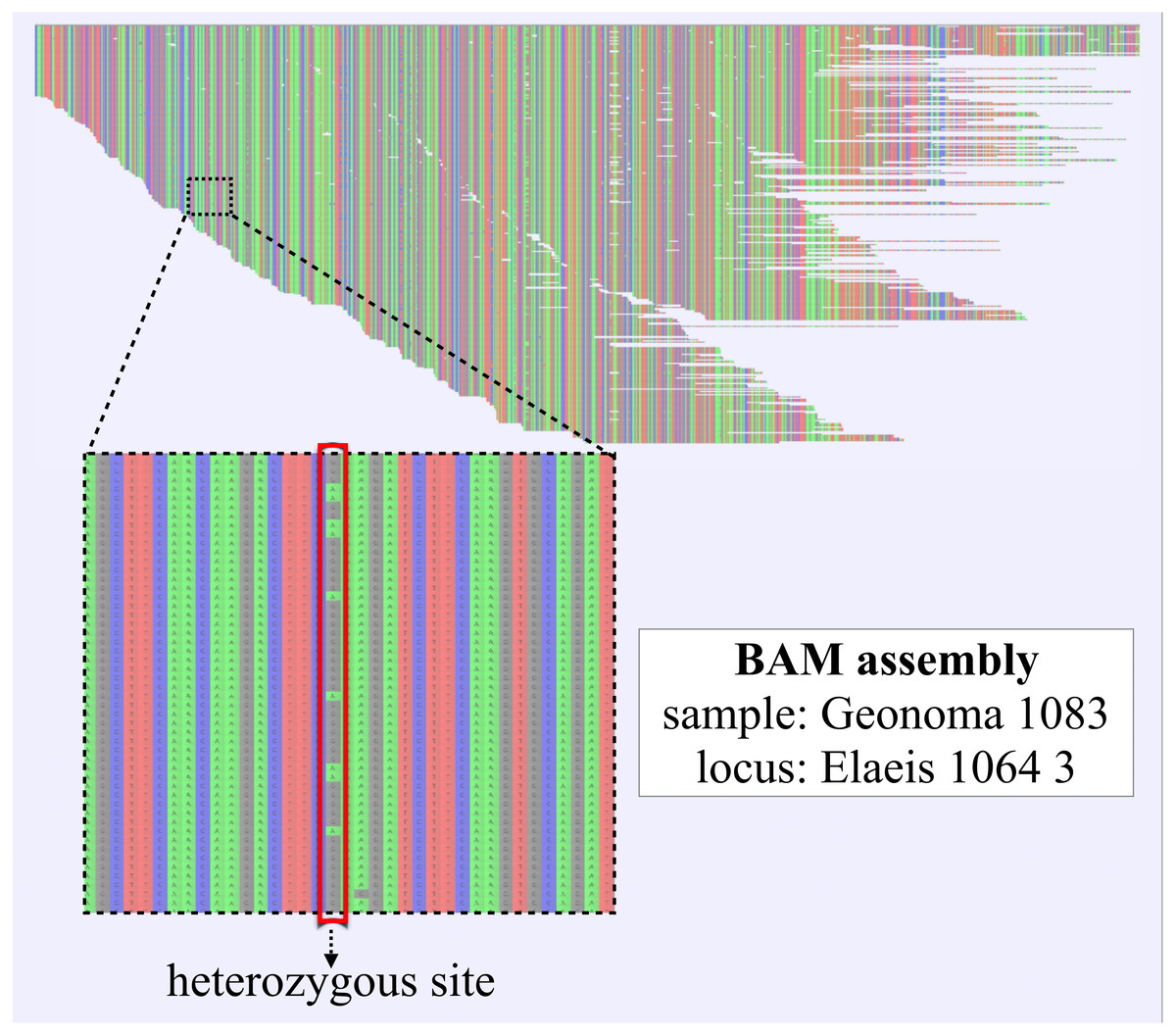
Fragmentation and decline of the amount of endogenous molecules are mainly due to depurination caused by hydrolysis while deamination of cytosines tends to occur at the 5′ ends, being translated in misincorporations C to T (C > T) at 5′ and G to A (G > A) at 3′ in the final sequence obtained from a double-stranded library ( Briggs et al., 2007). Degradation of the genetic material is due to factors such as temperature, pH, and processes as hydrolysis and oxidation that act on the biological sample through time. Endogenous ancient DNA (aDNA) molecules are often retrieved in low copy number, with the co-presence of possible exogenous contaminant DNA, and are characterized by high fragmentation and a typical pattern of damage at read termini. Genetic material recovered from ancient samples has particular characteristics due to degradations that occurred through time. A thorough functional annotation of detected and filtered mtDNA variants was also performed for a comprehensive evaluation of these ancient samples. Potential heteroplasmy levels were also estimated, although most variants were likely homoplasmic, and validated by data simulations, proving that new sequencing technologies and software are sensitive enough to detect partially mutated sites in ancient genomes and discriminate true variants from artifacts. Additionally, we provide guidelines to deal with possible artifact sources, including nuclear mitochondrial sequence (NumtS) contamination, an often-neglected issue in ancient mtDNA surveys. Through a fine-tuned filtering on variant allele sequencing features, we were able to accurately reconstruct nearly complete (>88%) mtDNA genome for almost all the analyzed samples (27 out of 30), depending on the degree of preservation and the sequencing throughput, and to get a reliable set of variants allowing haplogroup prediction. The pipeline includes several modules from well-established tools for aDNA analysis and a recently released variant caller, which was specifically conceived for mtDNA, applied for the first time to aDNA data. We implemented and applied a computational pipeline for mtDNA analysis to a dataset of 30 ancient human samples from an Iron Age necropolis in Polizzello (Sicily, Italy). In this regard, the assessment of the heteroplasmic fraction in ancient mtDNA has always been considered an unachievable goal due to the complexity in distinguishing true endogenous variants from artifacts. However, postmortem DNA damage and contamination make the data analysis difficult because of DNA fragmentation and nucleotide alterations. 2Dipartimento di Scienze e Tecnologie Biologiche, Chimiche e Farmaceutiche, Università degli Studi di Palermo, Palermo, ItalyĪncient DNA (aDNA) studies are frequently focused on the analysis of the mitochondrial DNA (mtDNA), which is much more abundant than the nuclear genome, hence can be better retrieved from ancient remains. 
1Dipartimento di Biologia, Università degli Studi di Firenze, Florence, Italy.Help me Obi-Wan Kenobi, you are my only hope.Maria Angela Diroma 1*† Alessandra Modi 1† Martina Lari 1 Luca Sineo 2 David Caramelli 1 Stefania Vai 1* And since I am running this job in parallel for multiple samples (each producing a unique final sequence), I want to find a solution more elegant than simply renaming the sequences 1 by 1. In my case I just have 1 sample, 1 sequence (the final consensus from the alignment) so I don't know where the name is fetched. Indeed most of the questions relating to this problem deal with multiple sequences inside an alignment (chromosomes, multiple samples, etc). I checked quite a few forums and the manuals of each software, but I can't figure out what to do. In this example, the fasta sequence in the SAMPLE1.fasta file should be named ">SAMPLE1", but it is always named ">reference". Seqtk seq -aQ64 -q13 -n N SAMPLE1_consensus.fastq > SAMPLE1.fasta Samtools mpileup -uf reference.fasta SAMPLE1_aln_sorted.bam | bcftools call -c | vcf2fq > SAMPLE1_consensus.fastq Samtools sort SAMPLE1_aln.bam -o SAMPLE1_aln_sorted.bam Samtools view -S -b SAMPLE1_aln.sam > SAMPLE1_aln.bam Minimap2 -ax sr reference.fasta SAMPLE1_trimmed.fastq > SAMPLE1_aln.sam Here is the script I use: # align reads to reference sequence with minimap2 I want to have the name of the fasta sequence to be the one of the original sequence file, not the reference. The problem is: my final consensus fasta sequence is always named after the reference sequence. I am assembling a virus genome from IonTorrent reads based on a reference fasta sequence, using a combination of Minimap2, Samtools, bcftools and such.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
